If you find this useful, please leave a comment at the end of the page.

SECTION A
1
Which sample contains the greatest number of molecules?
A
35.0 g of C2H2
B
45.0g of C2H6
C
60.0g of C4H10
D
100.0g of C6H6
To determine which sample contains the greatest number of molecules, we need to calculate the number of moles for each compound. The 1st step is to calculate the different molecular masses:
- C2H2: 2×12.0 + 2×1.0 = 26 g/mol
- C2H6: 2×12.0 + 6×1.0 = 30 g/mol
- C4H10: 4×12.0 + 10×1.0 = 58 g/mol
- C6H6: 6×12.0 + 6×1.0 = 78 g/mol
With these values, we obtain that the number of moles for each molecule is:
$$
\small
\mathrm{C_2H_2}:\ 35.0\ \text{g} \times \frac{1\ \text{mol}}{26\ \text{g}} = 1.35\ \text{mol}
$$
$$
\small
\mathrm{C_2H_6}:\ 45.0\ \text{g} \times \frac{1\ \text{mol}}{30\ \text{g}} = 1.50\ \text{mol}
$$
$$
\small
\mathrm{C_4H_{10}}:\ 60.0\ \text{g} \times \frac{1\ \text{mol}}{58\ \text{g}} = 1.03\ \text{mol}
$$
$$
\small
\mathrm{C_6H_6}:\ 100.0\ \text{g} \times \frac{1\ \text{mol}}{78\ \text{g}} = 1.28\ \text{mol}
$$
Therefore, the molecule with more moles (and therefore molecules) is C2H6, and the answer is B.
Your Answer: B
[1]
2
0.688g of an oxide of manganese is reduced by hydrogen gas to form manganese metal and 0.235g of water.
What is the formula of the oxide of manganese?
A
MnO
B
MnO2
C
Mn2O3
D
Mn3O4
The oxide of manganese (MnxOy) reacts with hydrogen gas to produce manganese metal and water, the unadjusted reaction is:
MnxOy + H2 → Mn + H2O
0.688 g 0.235 g
As each mole of H2O (18.0 g) has 1 mole of O (16.0 g), we can find that 0.235 g of water has:
$$ \small 0.235\ \text{g}\ \mathrm{H_2O} \times \frac{16.0\ \text{g}\ \mathrm{O}}{18.0\ \text{g}\ \mathrm{H_2O}} = 0.209\ \text{g}\ \mathrm{O} $$
All the O is provided by the oxide of manganese, we can calculate that the mass of manganese metal in the sample is
0.688 g – 0.209 g = 0.479 g of Mn
We can find the oxide metal as we calculate the empirical formulas:
| Atom | Atomic mass | Mass (or %) | Number of moles (mass/atomic mass) | Number of moles/lowest number of moles |
|---|---|---|---|---|
| O | 16.0 | 0.209 | 0.209÷16 = 0.01306 | 0.01306÷0.00872 = 1.50 |
| Mn | 54.9 | 0.479 | 0.479÷54.9 = 0.00872 | 0.00872÷0.00872 = 1.00 |
Multiplying by 2 both numbers, we have that the ratio O:Mn is 3:2, then, the manganese oxide is Mn2O3 and the answer is C.
Your Answer: C
[1]
3
How many hydrogen atoms are in 2.50g of pharmacolite, CaHAsO4•2H2O (Mr = 216.0)?
A
6.97 × 1021
B
2.09 × 1022
C
2.79 × 1022
D
3.48 × 1022
With 2.50 g of pharmacolite is:
$$\small 2.50\ \text{g pharmacolite} \times \frac{1\ \text{mol pharmacolite}}{216.0\ \text{g pharmacolite}} = 0.0116\ \text{mol pharmacolite}$$
As each mol of pharmacolite has 5 moles of hydrogen:
CaHAsO4·2H2O
We have:
$$\small 0.0116\ \text{mol pharmacolite}\ \times\ \frac{5\ \text{mol H}}{1\ \text{mol pharmacolite}} = 0.058\ \text{mol H}$$
Multiplying by Avogadro constant:
0.058 mol × 6.02 × 1023 = 3.49 × 1022
Accounting for rounding errors, the answer is D.
Your Answer: D
[1]
4
40.0 cm3 of 0.200 mol dm–3 HCl is added to 60.0 cm3 of 0.100 mol dm–3 NaOH.
What is the concentration of the resulting solution?
A
0.0200 mol dm–3 HCl and 0.0200 mol dm–3 NaCl
B
0.0200 mol dm–3 HCl and 0.0400 mol dm–3 NaCl
C
0.0200 mol dm–3 HCl and 0.0600 mol dm–3 NaCl
D
0.0600 mol dm–3 HCl and 0.0200 mol dm–3 NaCl
This is an acid base reaction:
HCl + NaOH→ NaCl + H2O
The 1st step is to calculate the initial number of moles of HCl and NaOH, using the concentrations and volumes given (for a brief calculation to spot the correct answer, we can ignore the units and work quickly with the calculator, remember that you are not marked for showing you work on this section).
Moles of HCl:
$$\small 40\ \text{cm}^3 \times \frac{0.2\ \text{mol}}{1\ \text{dm}^3} \times \left(\frac{1\ \text{dm}}{1\ \text{cm}}\right)^3 = 8 \times 10^{-3}\ \text{mol HCl}$$
Moles of NaOH:
$$\small 60\ \text{cm}^3 \times \frac{0.1\ \text{mol}}{1\ \text{dm}^3} \times \left(\frac{1\ \text{dm}}{1\ \text{cm}}\right)^3 = 6 \times 10^{-3}\ \text{mol NaOH}$$
We have an excess if HCl, leaving in the final solution:
8×10-3 – 6×10-3 = 2×10-3 moles of HCl
All the NaOH has been consumed to produce 6×10-3 moles of NaCl.
As the final volume (assuming that volumes are additive), we get 40 + 60 = 100 cm3, or 0.1 dm3. If we divide the moles by the 0.1, we get the following concentrations:
- HCl: 2×10-3÷ 0.1 = 2×10-2 mol dm-3
- NaOH: 6×10-3÷ 0.1 = 6×10-2 mol dm-3
Therefore, the answer is C.
Your Answer: C
[1]
5
Which compound has polar molecules?
A
HCN
B
BCl3
C
CO2
D
C2F4
HCN is a polar molecule because it has a linear structure, and there’s a significant electronegativity difference between hydrogen (H), carbon (C), and nitrogen (N). This creates a dipole moment.
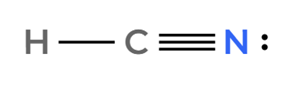
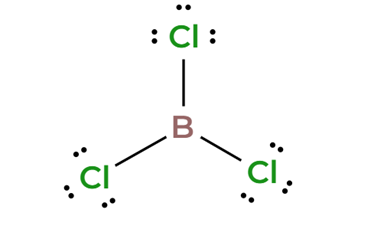
BCl3 is non-polar despite the individual B–Cl bonds being polar. The molecule’s trigonal planar geometry makes the dipole moments cancel out.
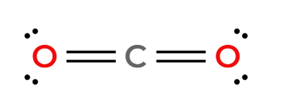
CO₂ has polar bonds, but its linear structure causes the dipole moments to cancel out, making the molecule non-polar.
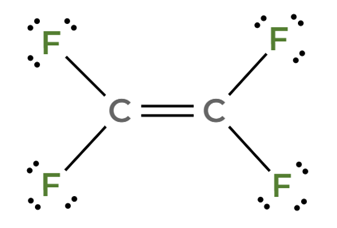
C₂F₄ is a planar molecule, where the dipole moments cancel each other.
Therefore, the answer is A.
Your Answer: A
[1]
6
Which element has the largest third ionisation energy?
A
Li
B
F
C
Ne
D
Na
The element with the largest third ionisation energy is the lithium. In neutral state it has 3 electrons. After removing two electrons, the third electron comes from the orbital 1s, which is very close to the nucleus, with no other electrons shielding the nucleus charge and it is strongly held.
A table with the different ionization energies can be found in Molar ionization energies of the elements – Wikipedia
Your Answer: A
[1]
7
The reaction below is first order with respect to reactant X.
X(aq) → products
When the initial concentration of X is 1.0 mol dm–3, the half-life is 16 minutes.
What is the half-life when the initial concentration of X is 2.0 mol dm–3?
A
2 minutes
B
4 minutes
C
8 minutes
D
16 minutes
Half-life, t1/2 , is the time taken for the concentration of a reactant to fall to half of its original value. For a reaction that is first-order with respect to the reactant, the half-life is independent of the initial concentration. This means that the half-life remains constant regardless of how much of the reactant is present initially.
In this case, since the reaction is first-order and the half-life at 1.0 mol dm⁻³ is 16 minutes, the half-life will also be 16 minutes when the initial concentration of X is 2.0 mol dm⁻³. The answer is D.
Your Answer: D
[1]
8
Which compound requires the most energy to convert one mole into its gaseous ions?
A
NaF
B
Na2O
C
MgF2
D
MgO
All the compounds listed are ionic solids. To break them into their gaseous ions, we need to overcome their lattice energies. The two key factors affecting lattice energy are:
- Ionic Charges: Higher charges on the cation and anion result in stronger electrostatic attraction and therefore higher lattice energy.
- Ionic Radii: Smaller ions can pack closer together, increasing the strength of the attraction. Larger ionic radii reduce lattice energy.
Since all the ions in the options are relatively small, the main factor here is the ionic charges:
- NaF is formed by Na+ and F–.
- Na2O is formed by Na+ and O2-.
- MgF is formed by Mg2+ and F–.
- MgO is formed by Mg2+ and O2-.
MgO has the highest charges (+2 on Mg2+ and −2 on O2−), giving it the strongest electrostatic attraction and the highest lattice energy. Therefore, MgO requires the most energy to convert one mole into gaseous ions. The answer is D.
Your Answer: D
[1]
9
The table below shows standard entropies, Sө

What is the standard entropy change, ∆Sө, in JKmol–1, for the formation of 1 mol of SO3(l) from SO2(g) and O2(g)?
A
–508
B
–254
C
+254
D
+508
Standard entropy is the absolute entropy (amount of disorder or randomness) of a substance at a standard state (in its normal physical state at 298 K (25°C) and 100 kPa (~1 atm)).
In the case of a chemical reaction, we can calculate the standard entropy change using:
ΔSөSystem = ΔSөProducts – ΔSөReactants
The adjusted reaction is:
SO2(g) + 1/2O2 → SO3
Replacing the values that we have:
ΔSөSystem = 96 – (248 + 204/2) = -254
The answer is B.
Your Answer: B
[1]
10
What is the percentage dissociation of a 0.015 mol dm–3 solution of methanoic acid, HCOOH (Ka = 1.60 × 10–4 mol dm–3)?
A
0.016%
B
1.1%
C
1.82%
D
10.3%
The dissociation reaction is:
HCOOH ⇄ H++HCOO−
The concentrations in equilibrium can be calculated (more details about these calculations in Chemical Equilibrium –):.
| [HCOOH] | [H+] | [HCOO−] | |
|---|---|---|---|
| Initial | 0.015 | 0 | 0 |
| Change | -x | x | x |
| Equilibrium | 0.015 – x | x | x |
Using the concentration in equilibrium and the dissociation constant, we have:
$$
\small \text{K} _\text{a} = \frac{\text{[HCOO}^- \text{][H}^+ \text{]}}{\text{[HCOOH]}}
$$
$$
\small 1.60 \times 10^{-4} = \frac{x^2}{0.015 – x}
$$
It is worth to assume that x << 0.015, in this case we can simplify the equation:
$$
\small 1.60 \times 10^{-4} = \frac{x^2}{0.015}
$$
$$
\small x = \sqrt{1.6 \times 10^{-4} \times 0.015} = 1.55 \times 10^{-3}
$$
The percentage of dissociation can be calculated as:
$$
\small \text{Percentage dissociation} = \frac{\text{Concentration of dissociated acid}}{\text{Initial concentration}} \times 100
$$
$$
\small \text{Percentage dissociation} = \frac{1.55 \times 10^{-3}}{0.015} \times 100 = 10.3\%
$$
The answer is D
If you don’t make the approximation that x << 0.015 and use the whole equation, the positive value that I get for the solution is x = 1.471×10-3 and the percentage of dissociation is 9.8%, the closer value that I get from those provided in the answer is 10.3% (answer D).
Your Answer: D
[1]
11
A student adds aqueous potassium carbonate to one test tube and aqueous silver nitrate to a second test tube.
The student adds dilute hydrochloric acid to each test tube.
Which row has the correct observations?
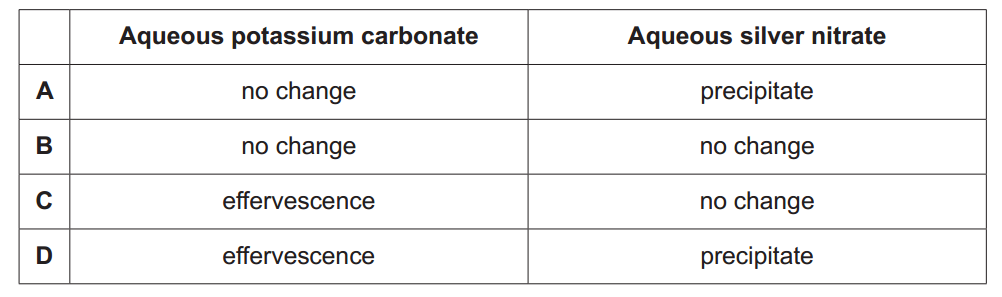
We have a reaction in each test tube.
In the test tube with potassium carbonate, we have:
K2CO3(aq) + 2HCl(aq) → 2KCl(aq) + CO2(g)↑ + H2O(l)
With the formation of CO2 gas, therefore the student observes effervescence.
In the test tube with aqueous silver nitrate, we have:
AgNO3(aq) + HCl(aq) → AgCl(s)↓ + HNO3(aq)
With the precipitation of silver nitrate, the student observes a precipitate.
The answer is D.
Your Answer: D
[1]
12
The equations show the electrode potentials of the half-cells used in a lithium-ion cell.
Eө/V
Li+ + e– ⇄ Li
–3.04
Li+ + CoO2 + e– ⇄ LiCoO2
+1.16
Which statement is correct in a lithium-ion cell?
A
The cell potential is 2.88V.
B
The reaction at the positive electrode is: LiCoO2 → Li+ + CoO2 + e–
C
The overall cell reaction is: Li + CoO2 → LiCoO2
D
The oxidation number of Co changes from +2 to +1.
For a reaction to occur spontaneously in a cell, the cell potential should be positive. To obtain a positive potential we invert the direction in the 1st semi reaction and add it to the 2nd:
Li → Li+ + e–
Eө = 3.04 V
Li+ + CoO2 + e– → LiCoO2
Eө = 1.16 V
__________________________________________________________________________
Li + CoO2 → LiCoO2
Eө = 4.20 V
We now check each option:
A:
The potential of the cell is 4.20 V, not 2.88 V. A is not correct.
B:
At the positive electrode (cathode), the reduction reaction occurs, the reaction in the positive electrode is Li+ + CoO2 + e– → LiCoO2. B is not correct.
C:
The overall reaction is the one indicated. C is correct.
D:
The oxidation number of the Co in the CoO2 is +4 (each O has an oxidation number of -2) and in the LiCoO2 is +3 (the Li has an oxidation number of +1 and each O has an oxidation number of -2). D is not correct.
Your Answer: C
[1]
13
Which substance(s) has/have induced dipole–dipole interactions (London forces) in the solid state?
1
C2H6
2
H2O
3
Si
A
1, 2 and 3
B
Only 1 and 2
C
Only 2 and 3
D
Only 1
Induced dipole-dipole interactions or London forces are weak intermolecular forces that occurs when the temporary uneven distribution of electrons in one molecule creates a temporary dipole, which then induces a similar dipole in a neighbouring molecule, leading to a weak attraction between them.
From the species given, the silicon is the only specie that forms a giant covalent bond, the other 2 are formed by discrete molecules and present London forces. In the case of water, the strongest intermolecular forces are hydrogen bond, but weaker dipole-dipole interactions are still present and therefore we cannot discard this answer. Therefore, the only specie that has no dipole-dipole interactions is silicon and the answer is B.
Your Answer: B
[1]
14
Which statement(s) is/are correct for the anti-cancer complex Pt(NH3)2Cl2?
1
It has bond angles of 90º.
2
The oxidation number of Pt is +4.
3
It forms both optical and cis-trans isomers.
A
1, 2 and 3
B
Only 1 and 2
C
Only 2 and 3
D
Only 1
The molecule is known as cisplatin. It is a square planar complex of platinum:
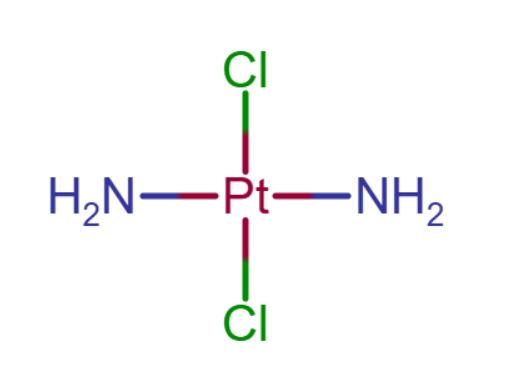
This structure present bond angles of 90°, therefore 1 is correct.
The oxidation number of Pt is +2, as each Cl contributes -1 and NH₃ is neutral. 2 is not correct.
The molecule exhibits cis-trans isomerism, with ligands of the same type positioned either adjacent to or opposite each other. However, it lacks chiral atoms, so optical isomers are not possible. 3 is not correct.
Only statement 1 is correct, and the answer is D.
Your Answer: D
[1]
15
Chlorine trifluoride can be decomposed into its elements forming the equilibrium mixture below.
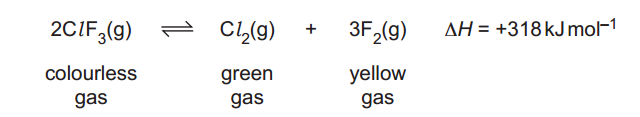
Which statement(s) is/are correct?
1
The decomposition is a redox reaction.
2
When the equilibrium mixture is cooled, the colour fades.
3
The decomposition has a negative entropy change.
A
1, 2 and 3
B
Only 1 and 2
C
Only 2 and 3
D
Only 1
The decomposition is a redox reaction that can be written as:
Oxidation: 2F– → F2 + 2e–
Reduction: 2Cl3+ + 6e– → Cl2
This is a redox reaction and statement 1 is correct. I explain more about the oxidation state of the chlorine in How to balance redox equations –.
The reaction is endothermic, a reduction in temperature displaces the equilibrium to the left, fading the colour. Statement 2 is correct.
In the decomposition, we pass from 2 moles of gases (from ClF3) to 4 moles of gases (1 from Cl2 and 3 from F2), therefore the entropy increases and it is not negative. Statement 3 is incorrect.
Only statements 1 and 2 are correct and the answer is B.
Your Answer: B
[1]
