Questions 3 to 5 are in OCR, AS Level Chemistry, 2023, H032/02 Depth in chemistry. Questions 3 to 5 –
If you find this useful, please leave a comment at the end of the page.
A link to a video of this paper can be found in https://youtu.be/tLww3fSvGH8

1
This question is about titanium (atomic number 22) and its compounds.
(a)
Titanium exists as a mixture of five isotopes.
A chemist analyses a sample of titanium using mass spectrometry.
The results are shown in the table below.
| Isotope | Abundance (%) |
|---|---|
| 46Ti | 8.30 |
| 47Ti | 7.40 |
| 48Ti | 73.70 |
| 49Ti | 5.40 |
| 50Ti | 5.20 |
(i)
Calculate the relative atomic mass of titanium in the sample.
Give your answer to 2 decimal places
To calculate the relative atomic mass of titanium in the sample, we use the weighted average of the isotopic masses based on their abundance percentages.
- We multiply each isotope’s mass number by its abundance percentage:
- We add all these products together.
- We divide this sum by the sum of all the percentages (that it should be 100, as the whole sample is titanium).
| Isotope | Abundance (%) | Mass number x abundance |
|---|---|---|
| 46Ti | 8.30 | 46×8.30=381.80 |
| 47Ti | 7.40 | 47×7.40=347.80 |
| 48Ti | 73.70 | 48×73.70=3,537.60 |
| 49Ti | 5.40 | 49×5.40=264.60 |
| 50Ti | 5.20 | 50×5.20=260.00 |
| Sum | 100 | 4791.80 |
4791.80/100=47.918
Rounded to 2 decimal places: 47.92.
relative atomic mass = ……………………47.92……………………………
[2]
(ii)
Complete the electron configuration of a titanium atom.
1s2 ………………………………………………………………………………………………………………..
As the atomic number (i.e. number of protons) is 22, the neutral atom has 22 electrons and the electronic configuration can be filled by using the energy level diagram with these 22 electrons.
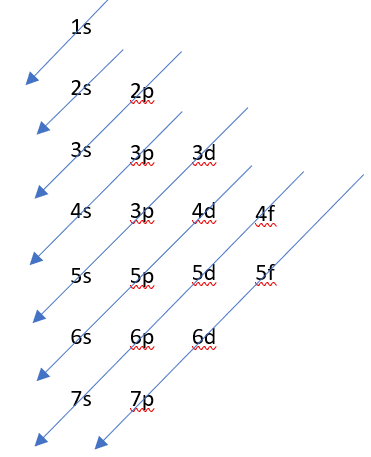
Alternatively, using the periodic table, we can see that the Ti is a transition metal of the 4th period, therefore, it is going to have the orbital 4s filled and we fill the orbital 3d with 2 electrons.
1s2 2s22p63s23p63d24s2………………………………………………………………………………………………………………..
[1]
(iii)
Complete the table to show the number of protons, neutrons and electrons in a 48Ti2+ ion.
| Protons | Neutrons | Electrons | |
|---|---|---|---|
| 48Ti2+ |
As the atomic number of titanium is 22, the number of protons is 22.
The problem asks for the isotope 48, then, number of protons + number of neutrons = 48, therefore, the number of neutrons is going to be 48 – 22 = 26.
Finally, since Ti2+ is the ion form, the atom has lost 2 electrons from the 22 in neutral state, therefore, the number of electrons is 22 – 2 = 20.
[1]
(b)
An ore of titanium contains impure TiO2.
Titanium is manufactured from TiO2 in a two-stage process.
| Stage 1 | TiO2 + 2C + 2Cl2 → TiCl4 + 2CO | Reaction 1.1 |
| Stage 2 | TiCl4 + 4Na → Ti + 4NaCl | Reaction 1.2 |
(i)
The common name for TiO2 is titanium dioxide.
What is the systematic name of TiO2?
Transition metal cations with multiple possible charges are labeled with Roman numerals in parentheses to indicate their specific charge. For example, titanium commonly has an oxidation state of +4, although other states like +3 can occur. In the TiO2, titanium is in the +4 oxidation state, hence the systematic name is Titanium (IV) oxide. For further reading, I found the Wikipedia page on IUPAC nomenclature of inorganic chemistry – Wikipedia particularly useful.
Titanium (IV) oxide.
[1]
(ii)
In Reaction 1.2, the percentage yield of titanium from TiCl4 is 72.0%.
Calculate the minimum mass, in kg, of sodium that is needed to produce 1.00kg of titanium.
Give your answer to 3 significant figures.
To obtain 1.00 kg of titanium with a 72.0% yield, we need to aim to get:
$$ \small 1.00 \times \frac{100}{72} = 1.39 \, \text{kg of Ti} $$
Using the molar mass of Ti as 47.9 g/mol, and changing from kg to g, we can get that:
$$ \small 1.390 \, \text{g Ti} \times \left( \frac{1 \, \text{mol Ti}}{47.9 \, \text{g}} \right) = 29 \, \text{mol Ti} $$
From the stoichiometry, we need 4 moles of Na for every mol of Ti, therefore, to get 29 mol of Ti, we need:
$$ \small 29 \, \text{mol Ti} \times \left( \frac{4 \, \text{mol Na}}{1 \, \text{mol Ti}} \right) = 116 \, \text{mol Na} $$
And using the molar mass of Na as 23.0 g/mol:
$$ \small 116 \, \text{mol Na} \times \left( \frac{23.0 \, \text{g Na}}{1 \, \text{mol Na}} \right) = 2668 \, \text{g Na} $$
Expressed in kg and with 3 significant figures we have 2.67 kg of Na.
mass of sodium = ……………………………………2.67……….. kg
[4]
(iii)
Reaction 1.2 produces a mixture of titanium and sodium chloride.
Suggest how titanium could be separated from this mixture at room temperature.
Explain your answer.
We can use that the NaCl is highly soluble in water, while Ti is not. Therefore, the titanium can be separated from the mixture by dissolving the sodium chloride in water, filtering the solid titanium, and then drying it.
[2]
2
This question is about some elements in Period 3 and compounds they form.
(a)
A student adds a small piece of calcium to a beaker containing an excess of water.
(i)
Construct the equation for the reaction and predict one observation that the student would make.
The reaction of group II metals calcium, strontium and barium with water produces the metal hydroxide and hydrogen gas:
M + 2H2O → M(OH)2 + H2
And the reactivity increases as we go down in the group.
Beryllium is no reactive with water at room temperature. However, it seems that it reacts with water vapour at 700°C (Reactions of the Group 2 elements with water)
Magnesium with cold water gives similar reactions than the Ca, Sr and Ba. Hot magnesium does react vigorously with water in
the form of steam to make magnesium oxide and hydrogen gas.
Mg(s) + H2O(l) → MgO(s) + H2(g)
Therefore, the answer is:
Reaction:
Ca + 2H2O → Ca(OH)2 + H2
Observation:
Any of the following observations would give you the 2nd mark:
- Formation of bubbles.
- The solid calcium dissolves.
- A white precipitate forms (this is accepted as correct in the marking scheme, although it depends on the proportions of calcium and water used by the student).
When asked about observations in an exam, focus on describing what you can actually see, rather than things you need to measure, like pH or temperature. For example, instead of mentioning changes in temperature (even though you might feel it by touching the beaker), stick to visible changes. This approach will help you avoid tricky answers during the exam.
[2]
(ii)
Suggest one difference that the student would observe in the reaction of barium with water compared to the reaction of calcium with water.
As the barium is more reactive than calcium, any of the observations mentioned above will be produced faster, i.e. one of the following will answer the question:
- The barium will produce more vigorous formation of bubbles.
- The solid dissolves faster.
- The white precipitate forms faster (as mentioned above, this is accepted as correct in the marking scheme, although it depends on the proportions of metal and water used by the student).
[1]
(b)
A student has a 5.00g mixture of sodium chloride, NaCl(s), and barium nitrate, Ba(NO3)2(s).
The student also has a solution of sodium sulfate, Na2SO4(aq).
The student uses the method below to determine the percentage by mass of NaCl(s) inta the mixture.
| Step 1 | Dissolve the 5.00g mixture in distilled water. |
| Step 2 | Add an excess of Na2SO4(aq) to the solution. A precipitate of barium sulfate forms. |
| Step 3 | Filter off the precipitate, wash with water, and dry. |
| Step 4 | Weigh the dried precipitate. |
The molar mass of barium sulfate is 233.4gmol–1.
(i)
Write an equation for the formation of barium sulfate in step 2.
Include state symbols.
When there’s a lot of information in a question, like this one, I start by reading through the entire question to get a general understanding of it. Then, I focus on the first part of the question and identify the relevant information needed to answer it, ignoring any other data for the time being. This way, I can concentrate on what’s important for each section and use the other information as needed for subsequent parts.
Information that I need:
- The student has a mixture of sodium chloride, NaCl, and barium nitrate, Ba(NO3)2.
- The mixture is dissolved in water.
- Excess of a solution sodium sulfate, Na2SO4 is added
- Barium sulfate precipitates.
To start with the reaction, I write the information that I have, bearing in mind that as the mixture has been dissolved in water, it is now in aqueous state:
NaCl(aq) + Ba(NO3)2(aq) + Na2SO4(aq) → BaSO4(s)
As the product is BaSO4, looking at the reactant, we can say that the NaCl has not taken part in the reaction and we can eliminate from the reaction:
Ba(NO3)2(aq) + Na2SO4(aq) → BaSO4(s)
Now, I write as ions any leftover from the reactants in the reaction and adjust all the species:
Ba(NO3)2(aq) + Na2SO4(aq) → BaSO4(s) + 2NO3– + 2Na+
And now, I match the ions to form ionic species, indicating that they are in aqueous form:
Ba(NO3)2(aq) + Na2SO4(aq) → BaSO4(s) + 2NaNO3(aq)
[2]
(ii)
The student obtains 3.28g of precipitate.
Calculate the percentage by mass of NaCl(s) in the 5.00g mixture.
Give your answer to 3 significant figures.
With the mass of the precipitate and its molar mass, we can calculate its number of moles:
$$ \small 3.28 \, \text{g BaSO}_4 \times \left( \frac{1 \, \text{mol BaSO}_4}{233.4 \, \text{g BaSO}_4} \right) = 0.014053 \, \text{mol BaSO}_4 $$
(Take at least 3 significant figures to minimize rounding errors).
With the stoichiometry of the reaction:
Ba(NO3)2(aq) + Na2SO4(aq) → BaSO4(s) + 2NaNO3(aq)
We have that 1 mol of BaSO4 requires 1 mol of Ba(NO3)2, therefore, the student started with 0.014053 mol of Ba(NO3)2. As the molar mass of the Ba(NO3)2 is:
137.3 + 2×(14 + 3×16) = 261.3 g/mol
We can calculate the mass of Ba(NO3)2 obtained by the student:
$$ \small 0.014053 \, \text{mol Ba(NO}_3\text{)}_2 \times \left( \frac{261.3 \, \text{g Ba(NO}_3\text{)}_2}{1 \, \text{mol Ba(NO}_3\text{)}_2} \right) = 3.672 \, \text{g Ba(NO}_3\text{)}_2 $$
As the mass of sample is 5.00 g, the mass of NaCl in the sample is:
5.00 – 3.672 = 1.328 g NaCl
Therefore, the percentage of NaCl is:
$$ \small \left( \frac{1.328}{5.00} \right) \times 100 = 26.56\% $$
And to 3 significant figures: 26.6%
percentage by mass of NaCl(s) = ……………………………………26.6………… %
[4]
(iii)
The student changes the method in 2(b).
In step 2, the student adds an excess of silver nitrate solution, AgNO3(aq), instead of Na2SO4(aq).
Explain whether this change would allow the student to determine the percentage by mass of NaCl(s) in the mixture.
The addition of silver nitrate instead of sodium sulfate will produce a precipitate of silver chloride, not barium sulfate. This will allow the student to determine the percentage of sodium chloride from the mass of the silver chloride precipitate.
(c)
The table below shows melting points and electrical conductivities of some elements in Period 3 and compounds they form.
| Substance | Magnesium sulfide, MgS | Aluminium, Al | Silicon, Si | Phosphorus trichloride, PCl3 |
|---|---|---|---|---|
| Melting point/°C | 2000 | 660 | 1414 | –94 |
| Electrical conductivity | – – | Good | Poor | – – |
| Type of lattice structure | Giant ………………. | ………………. ………………. | ………………. ………………. | ………………. ………………. |
(i)
Complete the table above to show the type of lattice structure of each substance.
Magnesium sulfide is a compound formed by a metal (Mg) and a non-metal (S), this combination produces ionic bonding between magnesium ions (Mg2+) and sulfide ions (S2-). The structure is giant ionic
Aluminium is a metal, therefore the structure is giant metal.
Silicon is a typical giant covalent structure.
Phosphorous chloride is a compound formed by 2 non-metals forming a single covalent (or simple molecular) structure.
| Substance | Magnesium sulfide, MgS | Aluminium, Al | Silicon, Si | Phosphorus trichloride, PCl3 |
|---|---|---|---|---|
| Melting point/°C | 2000 | 660 | 1414 | –94 |
| Electrical conductivity | – – | Good | Poor | – – |
| Type of lattice structure | Giant Ionic | Giant Metal | Giant Covalent | Simple Molecular |
[4]
(ii)
Explain the following:
- MgS has a higher melting point than PCl3.
- Al has a greater electrical conductivity than Si.
Melting points: In ionic bonds, like the magnesium sulfide, there are strong electrostatic attractions between the positively and negatively charged ions, requiring a lot of energy to break, resulting in a high melting point. On the other hand, in simple molecular structures, like the phosphorous trichloride, the molecules are held together by relatively weak Van Der Waals forces (intermolecular forces). These weak forces require less energy to overcome, resulting in a lower melting point.
Conductivity: The aluminium presents a metallic bonding with delocalized electrons. When a voltage is applied, the free electrons move easily through the metal, resulting in high electrical conductivity. On the other hand, in the silicon, each silicon atom is covalently bonded to four other silicon atoms forming a giant covalent structure. All valence electrons are involved in these covalent bonds, leaving no free electrons to conduct electricity.
[4]
Questions 3 to 5 are in OCR, AS Level Chemistry, 2023, H032/02 Depth in chemistry. Questions 3 to 5 –
